
Introduction
Quality failures in manufacturing don't just create defective products—they erode margins, damage customer relationships, and generate costly rework and waste. For manufacturers in competitive, specification-driven industries, the financial exposure is significant.
The Cost of Poor Quality (COPQ) consumes 15% to 20% of sales revenue for average organizations, with some reaching 40% of total operations.
The stakes extend beyond immediate scrap and rework costs. Late-stage defect discovery carries catastrophic financial risks, as evidenced by the Takata airbag crisis that resulted in $1 billion in criminal penalties and GM's ignition switch recall that cost $900 million to settle. Both cases illustrate what happens when systemic quality gaps go unaddressed until it's too late to contain the damage.
This article covers what quality control involves, the essential practices that underpin it, the methodologies available, and how to build a QC process that catches problems before they compound.
TLDR
- Quality control monitors and verifies products against defined standards throughout production—not just at the end of the line
- Effective QC combines clear standards, trained personnel, and real-time measurement to prevent defects—not just catch them
- Core methodologies include Six Sigma, Statistical Process Control, Total Quality Management, and Lean, each suited to different production environments
- A practical QC system follows five steps: define standards, select methods, train employees, conduct testing, and continuously improve
- Modern inline measurement technology makes QC proactive—catching deviations in real time before they become defects
What Is Quality Control in Manufacturing — and How Does It Differ from Quality Assurance?
Quality control consists of the operational activities used to detect and correct deviations from product specifications during and after production. It focuses on the product and what is produced, not solely on the system used to produce it.
The QA vs. QC Distinction
Understanding the difference between quality assurance and quality control is fundamental to building an effective quality management system.
According to the American Society for Quality (ASQ), quality assurance is "part of quality management focused on providing confidence that quality requirements will be fulfilled," while quality control is "part of quality management focused on fulfilling quality requirements."
In practice, the roles are distinct:
| Quality Assurance (QA) | Quality Control (QC) | |
|---|---|---|
| Orientation | Process-focused | Product-focused |
| Function | Preventive — defines standards, builds systems | Detective — verifies standards are being met |
| Question answered | "Are we following the right process?" | "Does the output meet requirements?" |
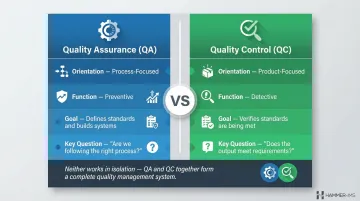
Neither discipline works well in isolation. QA sets the conditions for consistent output; QC confirms those conditions are delivering it. Together, they form the backbone of any functional quality management system.
ISO 9001: The Quality Standard Foundation
ISO 9001 represents the internationally recognized standard for quality management systems. According to the ISO Survey, there were 1,249,317 ISO 9001 certified sites worldwide in 2023 — a scale that reflects how widely the standard has been adopted across industries and geographies.
For manufacturers, aligning QC practices with ISO 9001 matters for three reasons:
- Regulatory compliance in sectors like medical devices and aerospace, where certification is often mandated
- Contractual requirements from customers who specify certified suppliers in procurement criteria
- Market access in regions where ISO 9001 certification serves as a baseline competitive requirement
Key ISO 9001 clauses directly relevant to QC include Clause 8.5.1 (Control of production), Clause 8.6 (Release of products), Clause 8.7 (Control of nonconforming outputs), and Clause 9.1 (Monitoring and measurement).
Why Quality Control Matters: The Real Cost of Getting It Wrong
Quality failures create costs that escalate sharply as defects move downstream. Total Quality Management describes this with the 1-10-100 Rule: every $1 spent on prevention and early detection saves $10 on appraisal and $100 in failure costs.
When defects escape detection, the financial impact multiplies:
- Internal failures: Scrap and rework disrupt production scheduling, inflate material consumption, and increase labour costs
- External failures: Defective products reaching customers damage brand reputation, trigger warranty claims, and—in the worst cases—result in recalls
- Operational disruption: Quality problems create bottlenecks, halt production lines, and require emergency root-cause investigations
In the food and consumer goods sector alone, label errors caused 45.5% of recalls in 2024, costing the industry an estimated $1.92 billion in direct expenses. Direct expenses are only part of the picture — lost customer trust and market share rarely appear on any balance sheet, but they compound long after the recall ends. Structured quality control practices are what prevent defects from reaching that stage.

Essential Quality Control Practices Every Manufacturer Should Follow
Define the Measurement Baseline First
No QC system functions without a clear, measurable definition of quality for each product. Manufacturers must establish specifications covering dimensions, weight, thickness, surface characteristics, or other critical parameters—and ensure these are objectively verifiable, not open to interpretation.
This means:
- Defining acceptable tolerance ranges for every critical parameter
- Establishing measurement methods and equipment calibration requirements
- Documenting specifications in formats that any trained inspector can apply consistently
- Reviewing and updating specifications when customer requirements or production capabilities change
Implement Inspection at Critical Control Points, Not Just at the End of the Line
End-of-line inspection alone is too late and too costly. Catching deviations early requires identifying critical control points (CCPs)—stages where a check can intercept a problem before it compounds downstream.
In discrete manufacturing, this is operationalised through Control Plans and Special Characteristics. Control Plans describe the system for controlling part and process quality against key engineering requirements. Special Characteristics are parameters that affect safety, compliance, fit, function, or subsequent processing—and receive priority attention during inspection.
Inline inspection technology detects defects almost immediately, preventing the need to re-sort production batches or scrap large quantities of suspect product. That early detection translates directly into less scrap, lower rework costs, and more predictable output.
Establish Clear Documentation and Traceability
Documentation creates the paper trail needed for audits, regulatory compliance, and root cause analysis. Without traceability from raw material intake to finished product dispatch, identifying the source of a quality failure becomes guesswork.
Essential documentation includes:
- Standard operating procedures (SOPs): inspection methods, equipment setup, and measurement frequency
- Inspection records: actual measurement data, timestamps, operator identification, and batch information
- Non-conformance reports: defect descriptions, severity, containment actions, and disposition decisions
- Calibration records: equipment accuracy logs confirming instruments remain within specified ranges
Train and Align All Personnel Involved in Production
Good documentation only works if the people using it are properly trained. Operators, technicians, and supervisors must understand what quality standards apply to their work, how to identify out-of-spec conditions, and what to do when deviations occur.
Poorly trained personnel are one of the most common sources of preventable quality failures. Training programmes should cover:
- Product specifications and acceptable tolerance ranges
- Proper use and care of measurement equipment
- Procedures for recording inspection data
- Response protocols when measurements fall outside specification
- Authority to stop production when critical defects are detected
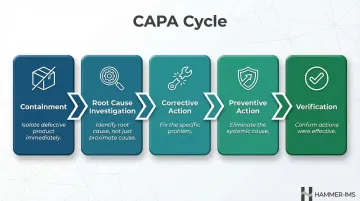
Close the Loop with Corrective and Preventive Action (CAPA)
CAPA represents the formal practice of addressing a detected defect (corrective action) and investigating and eliminating its root cause to prevent recurrence (preventive action). Tools like the 5 Whys and Fishbone (Ishikawa) diagrams help teams drill down to root causes rather than treating symptoms.
The CAPA process should include:
- Immediate containment of defective product
- Investigation to identify the root cause, not just the proximate cause
- Implementation of corrective actions to fix the specific problem
- Implementation of preventive actions to eliminate the systemic cause
- Verification that actions were effective through follow-up inspection
Key Quality Control Methodologies and When to Use Them
Statistical Process Control (SPC)
SPC is a real-time, data-driven method that monitors process variables (not just finished products) using control charts to detect process drift before defective output is produced. SPC applies statistical and analytical tools to control process inputs (independent variables), distinguishing it from inspection-based approaches that only measure outputs.
Best suited for: High-volume continuous production where even small process variations have significant quality impact—such as plastics extrusion, nonwovens production, or sheet manufacturing.
Key advantage: Catches problems at the process level before they create defects, enabling operators to adjust parameters proactively rather than reactively sorting good from bad product.
Six Sigma (DMAIC)
Six Sigma is a structured problem-solving methodology targeting near-zero defects. The numerical goal is 3.4 defects per million opportunities, achieved through the DMAIC framework: Define, Measure, Analyse, Improve, Control.
Best suited for: Situations where a specific recurring quality problem needs systematic root-cause investigation and permanent resolution, rather than ongoing process monitoring.
Key advantage: In 1987, Motorola spent $170 million on Six Sigma training for employees, which resulted in $2.2 billion in savings, demonstrating substantial return on investment when applied to chronic quality problems.
Total Quality Management (TQM)
TQM is an organisation-wide philosophy that embeds quality responsibility at every level—from shop floor operators to senior management. Unlike SPC or Six Sigma, which are applied to specific processes or problems, TQM is a cultural and systemic commitment to continuous improvement.
Best suited for: Organisations where leadership is willing to invest in long-term behavioural change and cross-functional quality ownership.
Key advantage: Creates sustainable quality culture rather than isolated improvement projects, but requires executive commitment and patience for cultural transformation.
Lean Quality Control and the 5S Framework
Lean's contribution to quality lies in eliminating the eight wastes: Defects, Overproduction, Waiting, Non-Utilisation of Talent, Transportation, Inventory, Motion, and Extra processing. By removing the conditions in which quality failures thrive, Lean creates inherently more stable processes.
The 5S workplace organisation framework (Sort, Set in order, Shine, Standardise, Sustain) supports consistent quality by creating orderly, standardised production environments where deviations are immediately visible.
Best suited for: Manufacturers dealing with quality issues rooted in disorganised workspaces, unclear procedures, or lack of standardisation.
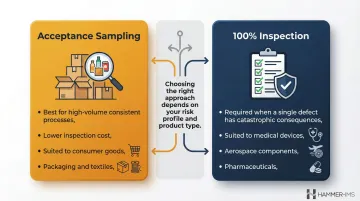
Statistical Quality Control (SQC) and Acceptance Sampling
SQC uses statistical tools—control charts, Pareto analysis, acceptance sampling—to evaluate production quality from representative samples rather than 100% inspection. ISO 2859-1 specifies acceptance sampling systems indexed by Acceptance Quality Limit (AQL).
Choosing between sampling and full inspection depends on risk profile and product type:
| Approach | Use When | Example Sectors |
|---|---|---|
| Acceptance sampling | High-volume, consistent processes where 100% inspection cost outweighs occasional defect risk | Consumer goods, packaging, textiles |
| 100% inspection | Even one faulty piece could have disastrous consequences | Medical devices, aerospace components, pharmaceuticals |
How to Build a QC Process: A Step-by-Step Approach
Step 1 — Define Quality Standards and Measurable Specifications
The first step is establishing what "acceptable quality" means in objective, measurable terms for each product. This involves reviewing customer requirements, regulatory specifications, and internal production capabilities.
Standards must be documented clearly enough that any trained inspector can apply them consistently. Vague requirements like "good surface finish" are unworkable; specifications must define measurable parameters such as surface roughness values, colour tolerances, or dimensional limits.
Step 2 — Select the Right QC Methods for Your Production Environment
Manufacturers should match their chosen QC methodology to their production context: volume, product type, risk profile, and available resources.
Consider these factors:
- Production volume: High-volume operations benefit from SPC and inline measurement; low-volume operations may rely more on batch sampling
- Product complexity: Simple products with few critical parameters need less elaborate QC systems than complex assemblies
- Risk profile: Safety-critical products justify more rigorous inspection than commodity items
- Available resources: Six Sigma requires trained personnel and time for DMAIC projects; simpler approaches may be more appropriate for smaller organisations
Step 3 — Train Employees and Establish Response Procedures
Consistent QC depends on people knowing exactly what to measure, how to measure it, and what to do when a measurement falls outside specification. Response procedures must be defined in advance, not improvised under pressure — covering who to notify, how to quarantine suspect product, and when to halt production. That means establishing:
- Clear authority structures for production hold decisions
- Containment procedures for out-of-spec product
- Escalation paths for quality issues requiring engineering or management involvement
- Documentation requirements for non-conformances
Step 4 — Monitor, Analyse Results, and Drive Continuous Improvement
A QC process requires ongoing attention. Regular analysis of data — defect rates, first-pass yield, scrap rates — surfaces trends before they become costly systemic failures.
Schedule periodic process audits and use QC data to drive structured improvement cycles. When data reveals a chronic problem, methodologies like DMAIC or Kaizen events give teams a framework to address root causes, not just symptoms.
The Role of Real-Time Measurement Technology in Modern QC
From Periodic Inspection to Continuous Monitoring
As manufacturing speeds increase and tolerances tighten, end-of-line or batch-sample inspection becomes insufficient. Real-time measurement systems positioned within the production line detect deviations at the moment they occur, enabling immediate corrective action before large quantities of out-of-spec material are produced.
McKinsey research shows that implementing smart inline quality control cuts response time to defects from an average of 50 minutes to under 5 minutes, whilst reducing defect rates from 3.2% to 0.3%. The implication is direct: manufacturers move from reacting to defects after the fact to preventing them mid-run.
Closed-Loop Production Control
Closed-loop systems take real-time measurement further: measurement data feeds directly back into the production process to automatically adjust parameters such as line speed, coating weight, or thickness, holding output within specification.
This approach reduces material waste, narrows production margins, and lowers the cost of quality over time. Hammer-IMS's M-Ray technology applies this principle directly: a contactless, non-nuclear inline measurement system delivering real-time thickness and basis weight data across textiles, nonwovens, plastics, and construction materials.
By feeding continuous measurements back to process controls, manufacturers can run precisely at specification limits without risking out-of-spec output. In artificial grass production, for example, M-Ray systems measure latex coating layers continuously, enabling automatic doctor blade adjustment to hold precise coating specs and reduce costs on expensive materials.

Enhanced Documentation and Traceability
Modern measurement technology delivers an additional benefit: continuous data logging creates a complete production record at no additional inspection labour cost.
That continuous record supports:
- Compliance audits — objective, timestamped data replaces manual sampling logs
- Customer quality reporting — documentation covering 100% of production, not just spot-checked batches
- Root cause investigations — precise data trails that pinpoint when and where a deviation began
For manufacturers in regulated industries or those facing tighter customer documentation requirements, this automated traceability adds value well beyond defect detection alone.
Frequently Asked Questions
What are the 4 types of quality control in manufacturing?
The four commonly recognised types are process control, product inspection, statistical sampling, and acceptance quality control (incoming material verification). Together, they cover every stage of production — from supplier inputs through to finished goods — and are most effective when used as a coordinated system rather than in isolation.
What are the 7 tools of quality control?
The classic 7 QC tools defined by Kaoru Ishikawa are: Cause-and-Effect (Fishbone) Diagram, Check Sheet, Control Chart, Histogram, Pareto Chart, Scatter Diagram, and Stratification (or Flow Chart). Each tool targets a different stage of problem analysis, from data collection through to root cause identification.
What are the 7 steps of a quality management system (QMS)?
Most QMS frameworks follow these seven steps: define quality policy and objectives, identify customer and regulatory requirements, design quality processes, implement and document procedures, monitor performance, conduct internal audits, and drive corrective action through management review. This sequence maps directly to ISO 9001:2015 clauses 4–10.
What are the 5 P's of quality assurance?
The 5 P's framework consists of: People (trained, quality-aware personnel), Processes (standardised, documented procedures), Products (goods meeting specifications), Plant/Equipment (maintained and calibrated tools and machinery), and Policies (organisational standards and quality commitments guiding all QA activity).
What is the difference between quality control and quality assurance in manufacturing?
Quality control is reactive and product-focused — it detects defects through inspection and testing during and after production. Quality assurance is proactive and process-focused — it prevents defects by designing and maintaining systems that consistently meet quality standards.


