
Quality failures in manufacturing are expensive. Cost of Poor Quality (COPQ) often consumes 15–20% of total sales through defects, rework, scrap, and warranty claims/ Strong inspection and Quality Control (QC) systems help prevent these losses by detecting problems early and improving process reliability.
This guide outlines essential inspection types, QC techniques, technology enablers, and best practices manufacturers can implement to reduce defects and improve consistency.
Key Takeaways
Integrating inspection and QC at every stage, from incoming materials to in-process and final inspections, helps detect defects early, reducing waste, rework, and costs.
Methods like visual checks, Automated Optical Inspection (AOI), NDT (X-ray/ultrasonic), dimensional measurements, and functional testing ensure products meet specifications and industry standards.
AI, automation, IoT sensors, and data analytics enhance accuracy, enable real-time monitoring, and support predictive quality management.
Key QC metrics, defect rate, First Pass Yield (FPY), COPQ, process capability, and customer feedback, provide actionable insights to optimize processes.
Documented inspections, employee training, Statistical Process Control (SPC), and proactive feedback loops foster a culture of quality, ensuring consistent, reliable production and customer satisfaction.
Types of Inspection
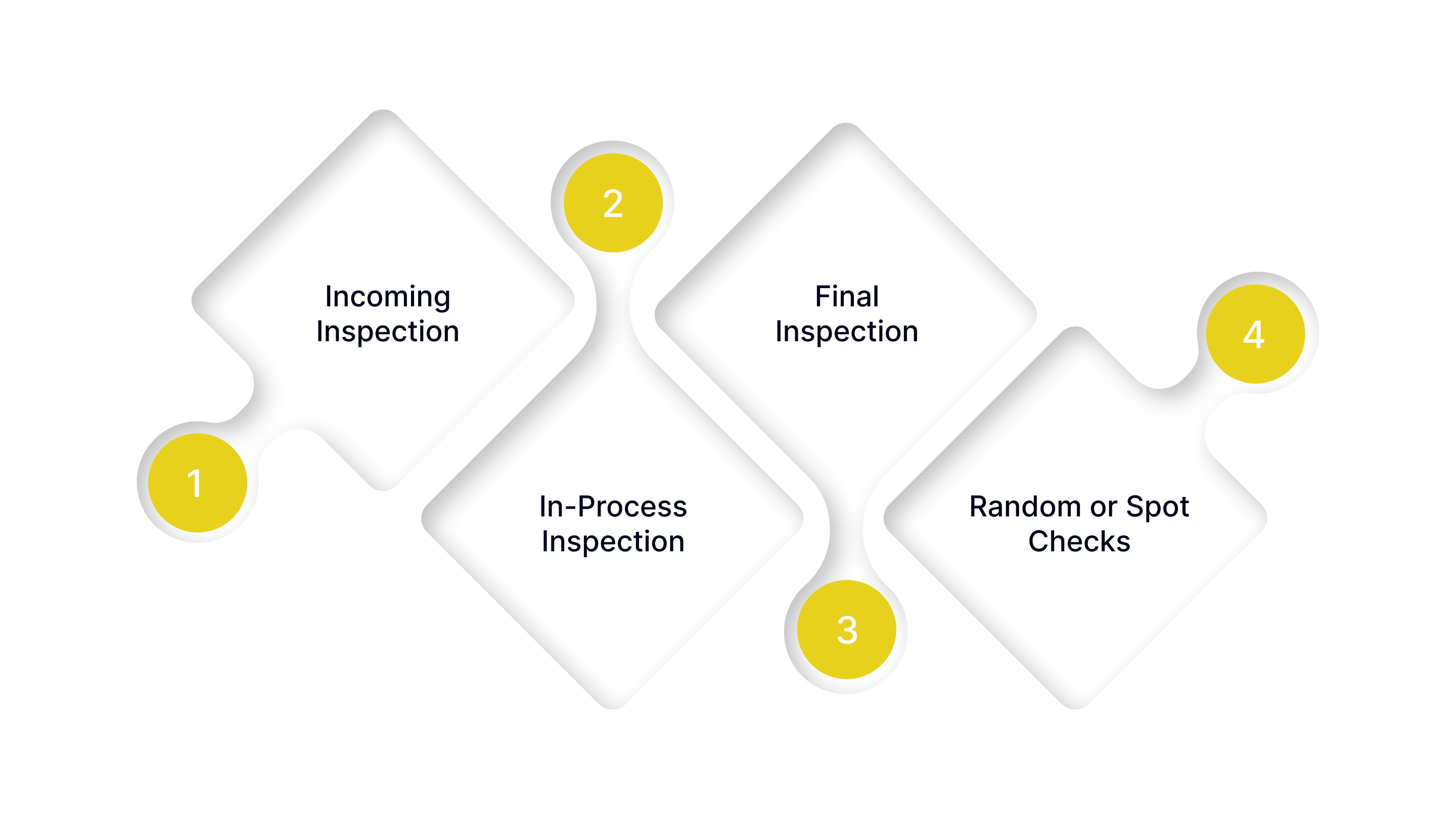
Inspection is a cornerstone of quality control in manufacturing. It ensures that materials, processes, and finished products meet the required standards and specifications. Manufacturers typically implement multiple types of inspection throughout the production cycle to catch defects early and maintain high-quality outputs.
1. Incoming Inspection
Purpose: To verify that raw materials, components, or subassemblies meet quality requirements before entering production.
When Conducted: Upon receipt from suppliers.
Methods / Tools: Visual checks, dimensional measurements, material testing, supplier documentation review.
Benefits: Prevents defective inputs from affecting production, reduces rework and scrap, and ensures supplier reliability.
2. In-Process Inspection
Purpose: To monitor and control quality during the manufacturing process.
When Conducted: At critical production stages or at regular intervals during production.
Methods / Tools: Automated sensors, sample testing, functional checks, and visual inspection.
Benefits: Detects defects early, reduces waste, ensures consistent production, and allows for quick corrective action.
3. Final Inspection
Purpose: To ensure finished products meet design specifications and customer expectations before shipment.
When Conducted: After the manufacturing process is complete.
Methods / Tools: Visual inspection, functional testing, dimensional verification, non-destructive testing (X-ray, ultrasonic).
Benefits: Guarantees that only compliant products reach the customer, reduces returns, warranty claims, and strengthens brand reputation.
4. Random or Spot Checks
Purpose: Quick verification of batch quality without inspecting every unit.
When Conducted: Intermittently during production or on selected samples.
Methods / Tools: Sampling, rapid tests, checklists.
Benefits: Provides an overview of overall production quality, saves time and resources while still detecting major issues.
5. Audit Inspection
Purpose: To evaluate adherence to manufacturing processes, regulatory standards, and internal quality protocols.
When Conducted: Periodically, quarterly, or annually.
Methods / Tools: Process audits, compliance reviews, documentation analysis.
Benefits: Identifies systemic issues, ensures compliance with standards such as Six Sigma, and drives continuous improvement.
Inspection Techniques and Methods
Manufacturers use a range of inspection techniques to ensure products meet quality standards. Selecting the right method depends on product type, complexity, material, and desired accuracy.
1. Visual Inspection
This is the simplest and most common inspection method. It relies on human observation to detect surface defects, misalignment, or assembly errors. Its applications include:
Electronics
Automotive components
Packaging
Surface finish checks
2. X-ray and Ultrasonic Testing
These are Non-Destructive Testing (NDT) methods that inspect internal structures without damaging the product.
X-ray Testing: Detects internal voids, cracks, or misalignments.
Ultrasonic Testing: Uses high-frequency sound waves to detect internal defects.
Their applications include:
Aerospace
Automotive
Pipelines
Castings
Electronics
3. Dimensional and Mechanical Inspection
This measures the physical dimensions and mechanical properties of components using tools like calipers, micrometers, Coordinate Measuring Machines (CMM), and hardness testers. Its applications include:
Precision engineering
Automotive parts
Metal fabrication
4. Functional Testing
It verifies that a product performs its intended function under real-world or simulated conditions. Its applications include:
Electronics (circuit testing)
Automotive (engine or system testing)
Consumer products
Medical devices
Best Practices in Quality Control
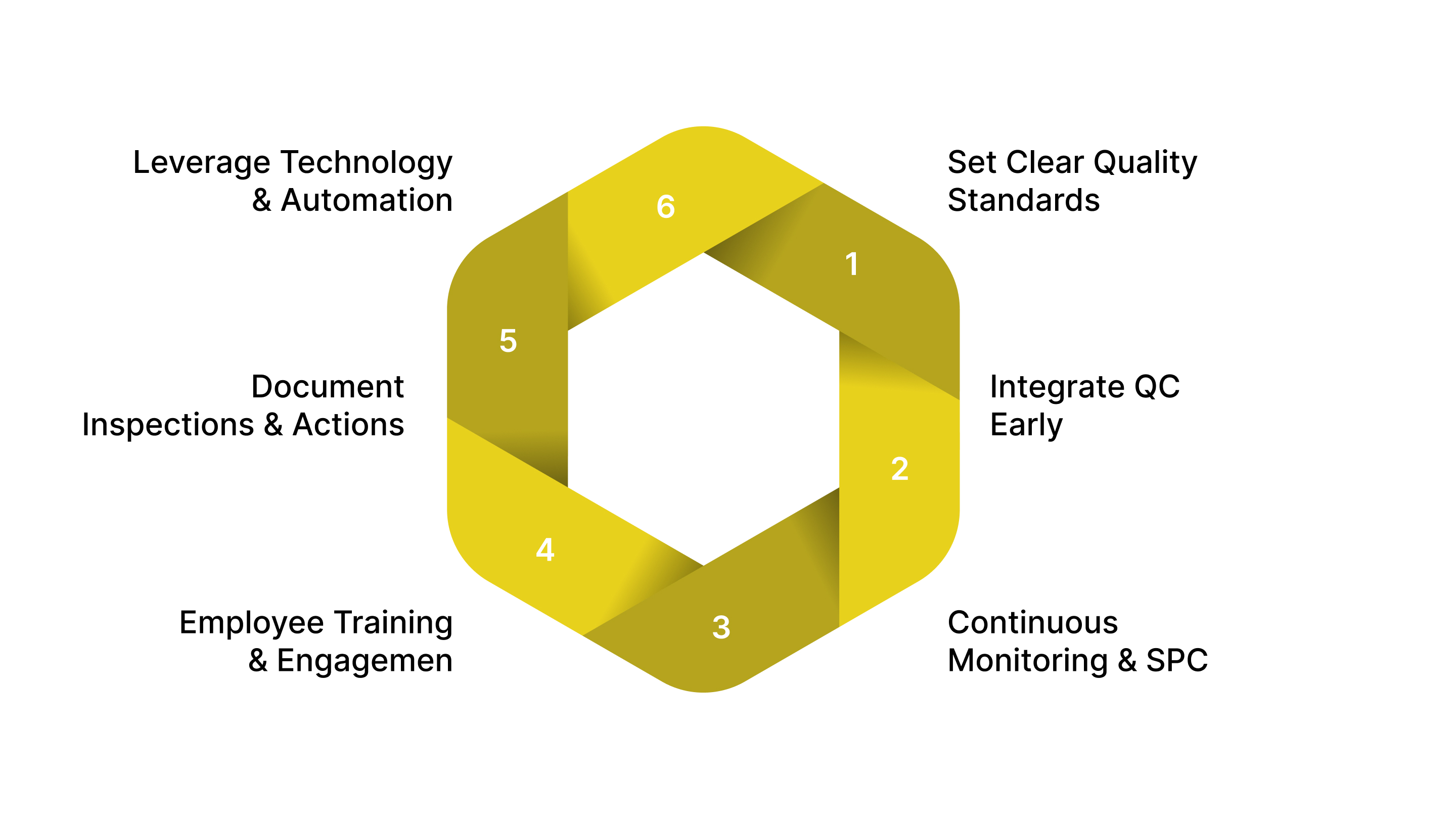
Implementing best practices in QC ensures consistent product quality, minimizes defects, and boosts overall manufacturing efficiency. Manufacturers can combine structured processes, advanced technologies, and employee engagement to build a robust QC system.
Establish Clear Quality Standards: Define explicit product specifications, tolerances, and inspection criteria. Adopt recognized frameworks such as Six Sigma,industry-specific standards. Ensure that all teams understand these standards to maintain consistency.
Integrate QC Early in Production: Conduct inspections at each critical stage rather than only at the end of production. Early detection of defects reduces scrap, rework, and production delays.
Use Continuous Monitoring and Statistical Process Control (SPC): Track quality metrics in real-time to detect trends and prevent defects before they escalate. Apply SPC charts to monitor variation and maintain process stability. Use predictive analytics and Internet of Things (IoT) sensors for proactive quality management.
Employee Training and Engagement: Train staff on QC procedures, inspection techniques, and the importance of quality standards. Encourage employees to identify potential quality issues and suggest improvements. Foster a quality-first culture, where every team member understands their role in maintaining product excellence.
Document Inspections and Corrective Actions: Record all inspection results, deviations, and corrective measures systematically. Documentation supports compliance audits and facilitates traceability.
Use Technology and Automation: Deploy automated inspection systems, AI-powered vision tools, and robotic testing to increase speed and accuracy. Use data analytics to identify root causes of defects and optimize processes. Integration with ERP or MES systems can streamline QC workflows and reporting.
Using Technology in Inspection and Quality Control
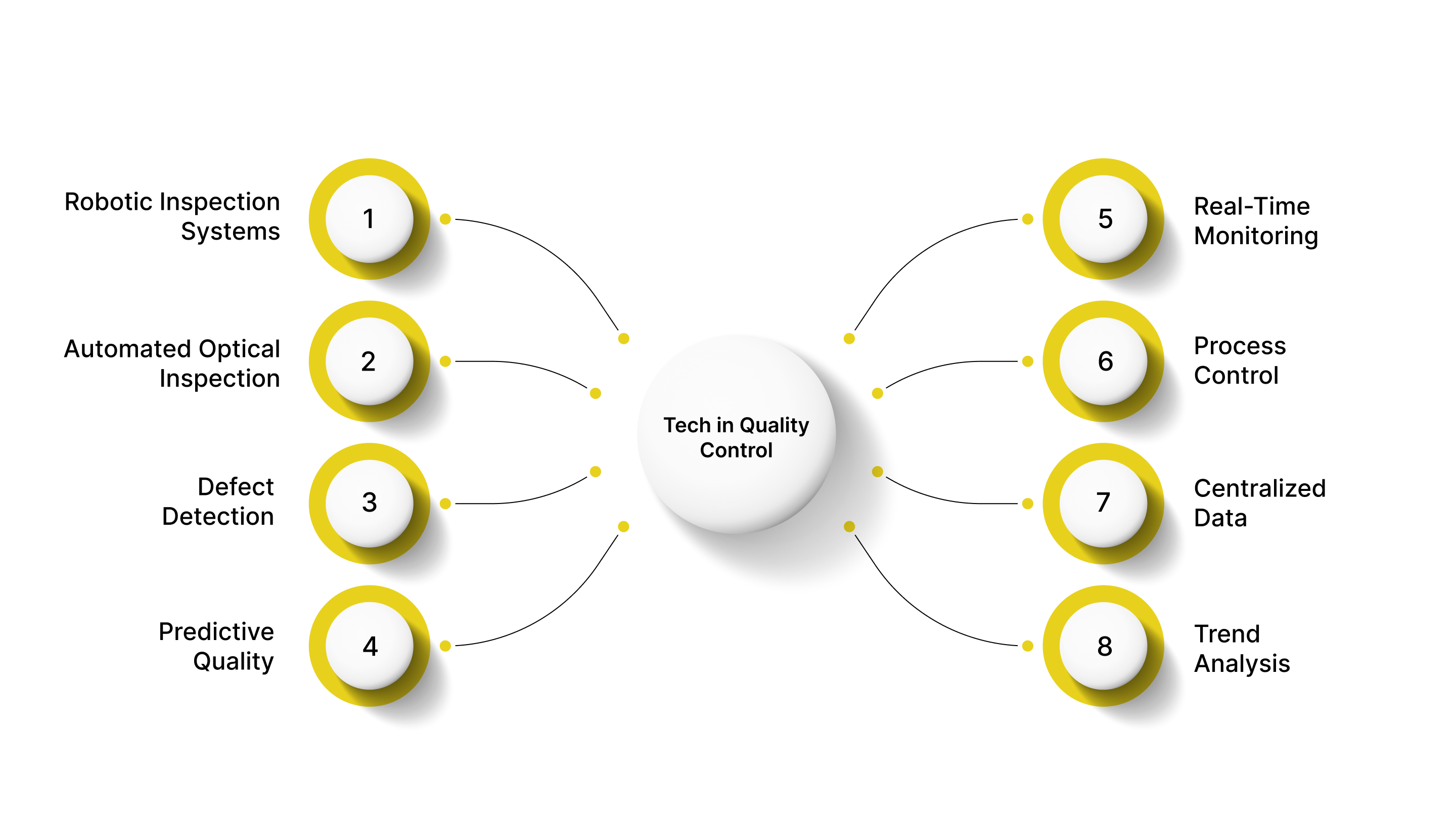
Advances in technology are transforming inspection and QC in manufacturing. By integrating automation, Artificial Intelligence (AI), and data analytics, manufacturers can enhance accuracy, reduce defects, and optimize production efficiency.
Robotic Inspection Systems: Automate repetitive inspection tasks such as visual checks, measurement, and assembly verification.
Automated Optical Inspection (AOI): AI-powered cameras detect micro-defects in electronics or complex assemblies with high speed and consistency.
Defect Detection: AI algorithms analyze images, sensor data, and production patterns to detect defects that humans might miss.
Predictive Quality: Machine learning models predict defects before they occur, enabling preventive actions.
Real-Time Monitoring: IoT sensors track environmental conditions, machine performance, and product parameters continuously.
Process Control: Data collected from sensors enables proactive adjustments to maintain quality standards.
Centralized Data: QC data from inspections, audits, and production processes can be integrated into dashboards for real-time insights.
Trend Analysis: Identify recurring defects, process bottlenecks, and improvement opportunities.
Implementing a Quality Control Plan in Manufacturing
A successful quality control system begins with a structured implementation plan. Manufacturers must adopt systematic processes that maintain product consistency while supporting operational efficiency.
Creating an Effective Quality Control Plan
Developing a QC plan starts with clear objectives and a detailed understanding of what must be measured at each stage of production. Teams should identify critical quality parameters and define how they will be inspected and monitored.
Key planning steps include:
Defining quality objectives and measurable metrics
Identifying critical control points
Establishing inspection and testing methods
Determining sampling frequencies
Setting acceptance criteria
Creating documentation and reporting procedures
Key Components of Manufacturing Quality Control
A robust QC system relies on several essential components that work together to ensure continuous product reliability.
Core elements include:
Quality specifications and standards
Inspection and testing protocols
Documentation and record-keeping systems
Non-conformance management
Corrective and preventive action (CAPA) procedures
Performance monitoring and reporting systems
Establishing Quality Standards and Specifications
Quality control is built on clearly defined, measurable standards that outline acceptable product characteristics. These standards must reflect customer needs, regulatory expectations, and manufacturing capabilities.
Steps in developing quality standards include:
Reviewing industry and regulatory requirements
Analyzing customer specifications
Assessing production capabilities and constraints
Documenting quality parameters
Defining measurement and validation methods
Employee Training for Quality Control
Well-trained employees are central to effective manufacturing QC. Training programs should equip personnel with the skills needed to perform inspections, document findings, and uphold quality requirements throughout production.
Training should cover:
QC fundamentals and procedures
Inspection techniques and measurement tools
Documentation requirements and reporting standards
Proper equipment operation
Root-cause analysis and problem-solving methods
Safety practices related to inspection and testing
Quality Control Documentation and Procedures
Comprehensive documentation supports consistency, audit readiness, and effective process execution. These documents guide day-to-day operations and provide evidence of compliance.
Essential documentation includes:
Standard Operating Procedures (SOPs)
Quality Control manuals
Inspection and test records
Training materials
Audit and compliance reports
Monitoring and Improving Quality Control Systems
Quality control systems must be continually assessed and refined. Regular reviews help identify gaps, improve performance, and ensure alignment with evolving production requirements.
Key monitoring activities include:
Routine performance reviews
Analysis of quality metrics and trends
Internal process audits
Employee feedback and improvement suggestions
Updating procedures and system enhancements
Effective QC implementation requires consistent commitment, well-defined procedures, trained teams, and continuous improvement. Regular evaluation of these elements ensures that the quality control plan remains efficient, compliant, and adaptable to changing manufacturing needs.
Quality Control (QC) vs. Quality Assurance (QA)
In manufacturing, understanding the distinction between Quality Control (QC) and Quality Assurance (QA) is essential for building robust quality management systems. Though often used interchangeably, they serve distinct purposes and complement each other.
Aspect | Quality Control (QC) | Quality Assurance (QA) |
|---|---|---|
Objective | Detect and fix defects | Prevent defects through process improvement |
Focus | Product | Process |
Approach | Reactive | Proactive |
Methods | Inspection, testing, measurement | Audits, process design, training, and continuous improvement |
Timing | After production or during in-process checks | During process planning and development |
Outcome | Fewer defective products | Consistent, reliable production processes |
Standards, Tolerances, and Specifications
Consistency in manufacturing quality depends heavily on well-defined standards, tolerances, and specifications. These elements provide a clear framework for both production processes and inspection activities, ensuring that every product meets customer and regulatory requirements.
Standards: Standards are formalized rules or guidelines that define how processes should be performed and how products should be evaluated. Examples include:
Six Sigma: A methodology focused on reducing defects and variability.
Industry-Specific Standards: Automotive (IATF 16949), Aerospace (AS9100), Electronics (IPC standards).
Tolerances: Tolerances specify the acceptable range of variation for a particular measurement, dimension, or characteristic of a product. Examples include:
A metal rod may have a length specification of 100 mm ± 0.5 mm.
Surface roughness tolerances for precision components in aerospace applications.
Specifications: Detailed descriptions of product requirements, including materials, dimensions, performance criteria, and testing methods. Examples include:
Electrical components specifying voltage, current, and tolerance ratings.
Medical devices require biocompatible materials and sterilization standards.
Key Metrics in Quality Control
Measuring and monitoring quality is essential for effective manufacturing. Key metrics help identify defects, assess process performance, and drive continuous improvement.
Defect Rate: The percentage of products that fail inspection or do not meet quality standards.
First Pass Yield (FPY): The percentage of products that pass all inspections without requiring rework.
Cost of Poor Quality (COPQ): Total cost associated with defects, including scrap, rework, warranty claims, and lost sales.
Process Capability (Cp / Cpk): Statistical measures of how well a manufacturing process produces output within specified limits.
Customer Returns / Complaints: Number of products returned or complaints received due to quality issues.
Common Challenges in Manufacturing Quality Control
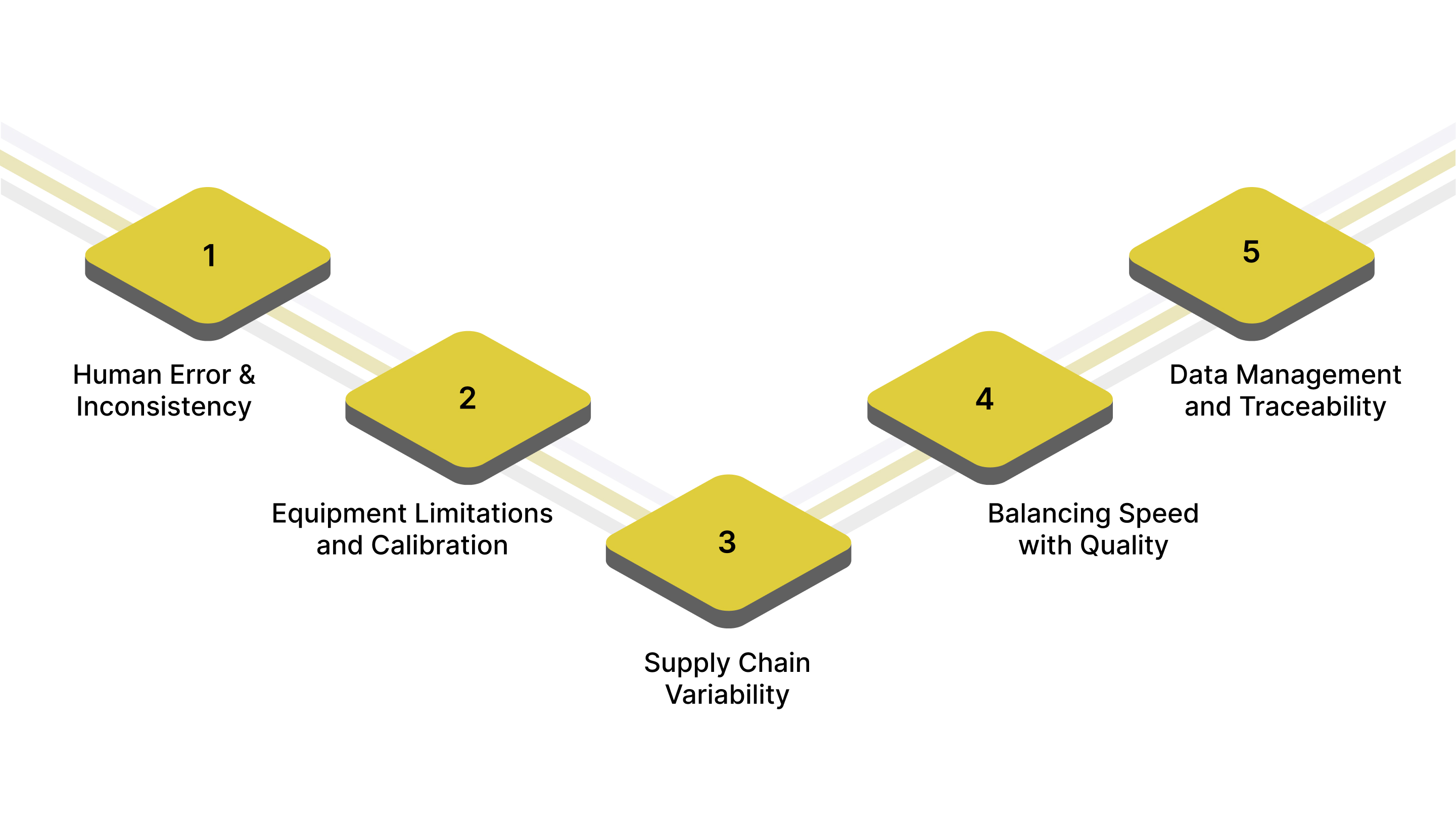
Despite advances in technology and process optimization, manufacturers face persistent challenges in maintaining consistent quality. Identifying and addressing these challenges is critical to prevent defects, reduce costs, and ensure customer satisfaction.
Human Error and Inconsistency: Manual inspections are prone to fatigue, oversight, and subjective judgment. To avoid this:
Implement automation for repetitive inspections.
Train employees regularly and establish Standard Operating Procedures (SOPs).
Encourage a culture of quality accountability.
Equipment Limitations and Calibration: Inspection tools and machinery can drift from calibration over time, leading to inaccurate measurements. To avoid this:
Establish regular maintenance and calibration schedules.
Use precision equipment suited to the required tolerances.
Monitor equipment performance with IoT-enabled sensors.
Supply Chain Variability: Quality of incoming materials or components can vary due to supplier inconsistencies or transportation conditions. To avoid this:
Conduct rigorous incoming inspections and supplier audits.
Develop strong supplier relationships and quality agreements.
Use predictive analytics to monitor material quality trends.
Balancing Speed with Quality: Production pressure for higher throughput may compromise inspection thoroughness or process adherence. To avoid this:
Integrate QC early in production rather than only at the end.
Automate inspections to maintain speed without sacrificing accuracy.
Monitor key metrics (FPY, defect rate, COPQ) to optimize the balance.
Data Management and Traceability: High-volume production generates large amounts of QC data, which can be difficult to manage effectively. To avoid this:
Implement digital dashboards and centralized Quality Management Systems (QMS).
Ensure proper documentation and traceability of inspection results.
Use analytics for actionable insights and continuous improvement.
Cost Considerations and ROI of Quality Control
Quality control requires meaningful investment, but when executed effectively, it delivers strong financial and operational returns. Understanding these cost factors helps manufacturers make informed, strategic decisions about QC improvements.
Cost-Effective Quality Control Implementation
Manufacturers must balance quality goals with budget constraints. A strategic, phased approach ensures that QC systems strengthen product reliability without inflating operational costs.
Key cost components include:
Investments in inspection equipment and automation
Training and upskilling of personnel
Staffing and resource allocation
Maintenance and calibration of tools
Documentation and quality management systems
Calculating the ROI of Quality Control
Evaluating ROI helps justify QC spending by quantifying the benefits of quality initiatives. These calculations typically consider both direct savings and long-term operational gains.
Major ROI drivers include:
Reduced scrap, rework, and waste
Fewer warranty claims and returns
Higher customer satisfaction and retention
Improved throughput and production efficiency
Lower overall inspection costs
Maintaining the Right Quality–Cost Balance
Achieving an optimal balance between quality and cost requires analyzing processes, risk levels, and customer expectations. This ensures that QC efforts remain effective and sustainable.
Factors that influence this balance include:
Prevention costs (training, process improvements)
Appraisal costs (inspection and testing)
Internal failure costs (scrap, rework)
External failure costs (returns, warranty claims)
Customer satisfaction and reputation impact
Hammer‑IMS: Enhancing Quality Control Through Advanced Inline Inspection
Maintaining consistent quality in modern manufacturing requires innovative inspection solutions, and Hammer‑IMS exemplifies how technology can transform quality control. Their inline measurement systems allow manufacturers to monitor key product parameters continuously, ensuring defects are detected immediately and standards are consistently met.
Precision Measurement Across Materials: Hammer‑IMS systems provide accurate monitoring of thickness, basis-weight, and supporting QC for plastics, textiles, battery films, and more.
Integration With Production Lines: Inline systems enable real-time quality verification without interrupting manufacturing, allowing faster corrective actions and minimizing waste.
Data-Driven Quality Decisions: Continuous data collection supports Statistical Process Control (SPC), trend analysis, and predictive maintenance, empowering manufacturers to improve processes proactively.
For manufacturers aiming to strengthen inspection protocols and ensure reliable product quality, Hammer‑IMS showcases how advanced technology can complement traditional QC practices. Book a demo to see how inline inspection can elevate your quality control processes.
Conclusion
Inspection and quality control are critical to ensuring consistent, high-quality manufacturing outcomes. By establishing clear standards, tolerances, and specifications, and employing techniques ranging from visual inspection to AI-powered automated systems, manufacturers can detect defects early and reduce waste.
Best practices, as mentioned above, enable proactive quality management. While challenges exist, combining human expertise with advanced tools and data-driven insights ensures operational efficiency, regulatory compliance, and customer satisfaction, turning quality control into a strategic advantage.
Take Your Quality Control to the Next Level.
Ensuring consistent, defect-free products requires a combination of best practices, skilled teams, and advanced inspection technologies. Hammer‑IMS and similar modern solutions demonstrate how inline, real-time quality monitoring can elevate your manufacturing processes, reduce waste, and improve overall efficiency.
Ready to enhance your inspection and quality control systems? Book a demo with Hammer‑IMS today and discover how precision, data-driven insights, and automation can transform your production line.
FAQs
1. How does preventive maintenance impact quality control?
Preventive maintenance keeps machinery calibrated and functioning properly, reducing the risk of defects caused by equipment wear or misalignment.
2. What role does supplier quality management play in QC?
Managing supplier quality ensures that raw materials and components consistently meet standards, reducing defects downstream in the manufacturing process.
3. Can Virtual Reality (VR) or Augmented Reality (AR) be used in quality inspections?
Yes, VR/AR can assist in training inspectors, visualizing defects, and simulating inspection procedures without disrupting production lines.
4. How do regulatory requirements affect inspection practices?
Industries like aerospace, automotive, and medical devices have strict regulatory standards that dictate inspection frequency, methods, and documentation.
5. What is the difference between destructive and non-destructive testing?
Destructive testing involves analyzing a sample until failure, while non-destructive testing inspects products without damaging them, preserving usability.
6. How can small manufacturers adopt advanced QC technologies cost-effectively
They can start with targeted automation for high-impact inspection points, leverage cloud-based analytics, or use shared inspection services to minimize upfront costs.
7. What is the role of continuous feedback loops in QC improvement?
Continuous feedback loops from inspection data help identify process bottlenecks, guide corrective actions, and support ongoing quality improvement initiatives.


